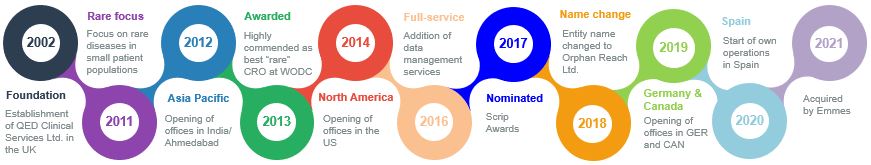
Orphan Reach is the result of 19 years of international trial experience which has started as QED Clinical Services and was initially established as a functional service provider for the pharmaceutical industry. QED quickly evolved into a niche CRO known for rescuing clinical studies from other CROs where patient recruitment and retention were the main challenges. Many of these projects have been conducted in smaller patient populations, particularly in rare diseases where larger service providers struggled to find the optimal development strategy.
Our team discovered that in many cases the patient’s interests were simply ignored and trials in rare diseases were handled in the same fashion than in more common disease areas. The consequences could ultimately be devastating not only for the Sponsor but also – even more concerning – for the patient desperately waiting for new therapies entering the market. This observation combined with an environment of increased orphan product development activity has inspired us to tackle the challenges in rare diseases and to create Orphan Reach, a dedicated CRO purely focused on unmet medical need.
Our team discovered that in many cases the patient’s interests were simply ignored and trials in rare diseases were handled in the same fashion than in more common disease areas. The consequences could ultimately be devastating not only for the Sponsor but also – even more concerning – for the patient desperately waiting for new therapies entering the market. This observation combined with an environment of increased orphan product development activity has inspired us to tackle the challenges in rare diseases and to create Orphan Reach, a dedicated CRO purely focused on unmet medical need.
